May 25, 2023
With the use of electric vehicles (EVs) and grid-scale energy storage systems on the rise, the need to explore alternatives to lithium-ion batteries (LIBs) has never been greater. One such replacement could be calcium (Ca) metal batteries. A prototype of a rechargeable battery utilizing Ca metal has been created by a research team, achieving an impressive milestone of 500 cycles of charge and discharge.
As the fifth most abundant element in earth's crust, calcium is widely available, inexpensive, and has higher energy density potential than LIBs. Its properties also help accelerate ion transport and diffusion in electrolytes and cathode materials, giving it an edge over other LIB alternatives such as magnesium and zinc.
But many hurdles still need to be overcome regarding Ca metal batteries' commercial viability. The lack of an efficient electrolyte and the absence of cathode materials with sufficient Ca2+ storage capabilities have proved to be the main stumbling blocks.
In 2021, some members of the current research group provided a solution to the former problem when they realized a new fluorine-free calcium (Ca) electrolyte based on a hydrogen (monocarborane) cluster. The electrolyte demonstrated markedly improved electrochemical performances, such as high conductivity and high electrochemical stabilities.
“For our current research, we tested the long-term operation of a Ca metal battery with a copper sulfide (CuS) nanoparticle/carbon composite cathode and a hydride-based electrolyte,” stated Kazuaki Kisu, assistant professor at Tohoku University's Institute for Materials Research (IMR).
Also, a natural mineral, CuS, has favorable electrochemical properties. Its layered structure enables it to store a variety of cations, including lithium, sodium, and magnesium. It has a significant theoretical capacity of 560 mAh g-1 – which is two to three times higher than present cathode materials for lithium-ion batteries.
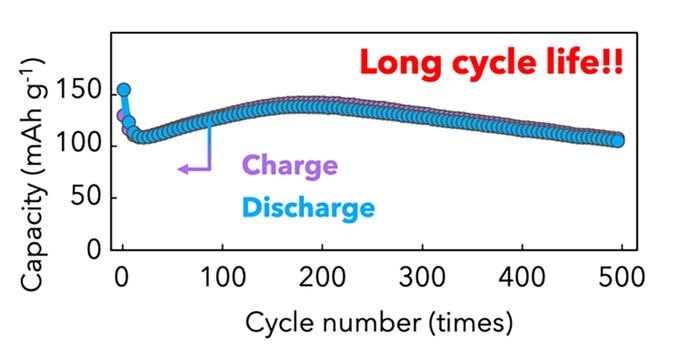
Kisu and his colleagues created a cathode capable of storing large amounts of calcium ions through nanoparticulation and compositing with carbon materials. When employed with the hydride-type electrolyte, they produce a battery with a highly stable cycling performance. The prototype battery maintained 92% capacity retention over 500 cycles based on the capacity of the 10th cycle.
The group is confident that their breakthrough will help advance research into cathode materials for Ca-based batteries. “Our study confirms the feasibility of Ca metal anodes for long-term operations, and we are hopeful the results will expedite the development of Ca metal batteries,” stated Kisu.
About the Author(s)
You May Also Like





