A Berkeley Lab-led team designs next-gen batteries at the atomic level that are less dependent of critical metals difficult to source do to supply chain issues. Meanwhile, a research team at the Technical University of Munich (TUM) has discovered a material class with above-average conductivity.
Scientists are still trying to find an ideal battery that is not only lighter but also safer, performs better, and lasts longer. It is very possible that the battery of the future will be a solid-state battery since the electrolyte in it is a solid substance, which can neither leak nor burn, and can make it lighter. Here are a couple of new discoveries from two different research teams:
Berkeley National Laboratory’s solid state battery:
A team from Lawrence Berkeley National Laboratory (Berkeley Lab) and Florida State University—supported by the DOE Vehicle Technologies Office—has designed a new blueprint for solid-state batteries that are less dependent on specific chemical elements, particularly critical metals that are challenging to source due to supply chain issues. Their work, reported recently in the journal Science, could advance solid-state batteries that are efficient and affordable.
Touted for their high energy density and superior safety, solid-state batteries could be a game-changer for the electric car industry. But developing one that is affordable and also conductive enough to power a car for hundreds of miles on a single charge has long been a challenging hurdle to overcome.
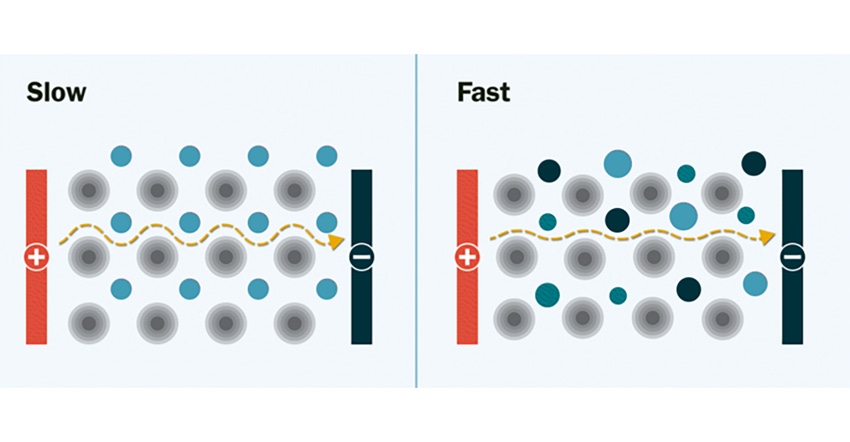
“With our new approach to solid-state batteries, you do not have to give up affordability for performance. Our work is the first to solve this problem by designing a solid electrolyte with not just one metal but with a team of affordable metals," stated co-first author Yan Zeng, a staff scientist in Berkeley Lab’s Materials Sciences Division.
In a lithium-ion battery, the electrolyte works like a transfer hub where lithium ions move with electric charge to either power a device or recharge the battery. Like other batteries, solid-state batteries store energy and then release it to power devices. But rather than liquid or polymer gel electrolytes found in lithium-ion batteries, they use a solid electrolyte.
Government, research, and academia have heavily invested in the research and development of solid-state batteries because the liquid electrolytes designed for many commercial batteries are more prone to overheating, fire, and loss of charge. However, many of the solid-state batteries constructed thus far are based on specific types of metals that are expensive and not available in large quantities. Some aren’t found at all in the United States.
For the current study, Zeng—along with Bin Ouyang, an assistant professor in chemistry and biochemistry at Florida State University and senior author Gerbrand Ceder, a Berkeley Lab faculty senior scientist and UC Berkeley professor of materials science and engineering—demonstrated a new type of solid electrolyte consisting of a mix of various metal elements. Zeng and Ouyang first developed the idea for this work while finishing their postdoctoral research at Berkeley Lab and UC Berkeley under the supervision of Ceder.
The new materials could result in a more conductive solid electrolyte that is less dependent on a large quantity of an individual element. In experiments at Berkeley Lab and UC Berkeley, the researchers demonstrated the new solid electrolyte by synthesizing and testing several lithium-ion and sodium-ion materials with multiple mixed-metals.
They observed that the new multi-metal materials performed better than expected, displaying an ionic conductivity several orders of magnitude faster than the single-metal materials. Ionic conductivity is a measurement of how quickly lithium ions move to conduct electric charge.
The researchers theorize that mixing many different types of metals together creates new pathways—much like the addition of expressways on a congested highway—through which lithium ions can move quickly through the electrolyte. Without these pathways, the movement of lithium ions would be slow and limited when they travel through the electrolyte from one end of the battery to the other, Zeng explained.
To validate candidates for the multi-metal design, the researchers performed advanced theoretical calculations based on a method called density-functional theory on supercomputers at the National Energy Research Scientific Computing Center (NERSC). Using scanning transmission electron microscopes (STEM) at the Molecular Foundry, the researchers confirmed that each electrolyte is made of only one type of material – what scientists call a “single phase” – with unusual distortions giving rise to the new ion transport pathways in its crystal structure.
The discovery enables new opportunities to design next-generation ionic conductors. The next step in this research is to apply the new approach that Zeng has developed with Ceder at Berkeley Lab to further explore and discover novel solid electrolyte materials that can improve battery performance even further. This work represents one of the many ways in which experts at the Berkeley Lab Energy Storage Center are working to enable the nation’s transition to a clean, affordable, and resilient energy future.
Technical University of Munich’s solid-state battery:
The research team at the Technical University of Munich (TUM) has discovered a material class with above-average conductivity. This is a decisive step forward in the development of high-performance solid-state batteries. Investigations conducted at the Research Neutron Source Heinz Maier-Leibnitz (FRM II) made an essential contribution to the discovery.
According to Prof. Thomas Fässler of the TUM Professorship for Inorganic Chemistry with Focus on New Materials, the solid-state electrolytes available up to now, mostly oxidic ceramics or compounds based on sulfur, have proven unable to completely meet expectations. Together with his team and in close cooperation with TUMint·Energy Research GmbH, he is looking for more efficient electrolytes. "The problem is that lithium ions only diffuse slowly through solid materials. Our objective was to better understand ion transport and then to use this knowledge to increase conductivity," Fässler stated.
The result of their efforts is a crystalline powder which is an above-average conductor of lithium ions. It contains no sulfur, but rather phosphorus, aluminum, and a comparatively high proportion of lithium. Laboratory measurements have shown that this previously overlooked substance class has a high level of conductivity. Within a very short period of time the chemists successfully created about a dozen new, related compounds, which contain for example silicon or tin instead of aluminum. This broad new material basis makes it possible to quickly optimize material properties.
Fässler explained that to see the good ion conductivity of these materials, the processes which take place inside the crystals must be rendered visible. "But that's not possible with normal laboratory equipment, because the lithium atoms are very light. As a result they cannot be localized exactly using X-ray radiation,” he continued.
The solution to see the processes which take place inside the crystals is the use of neutron beams. "The neutrons we have from the research reactor make it possible to find even the lightest of atoms. This is because the neutrons interact with the nuclei of the atoms and not with the atomic shell, as is the case with X-ray radiation," stated Dr. Anatoliy Senyshyn, who supervises the powder diffractometer at the FRM II, which was used to analyze the new electrolyte material: "In the past we had already investigated a variety of members of the new and diverse family of solid lithium ion conductors. We can use the neutron diffraction to visualize how the ions use free space in the crystal lattice to move," Senyshyn continued.

Dr. Anatoliy Senyshyn at the SPODI powder diffractometer at FRM II.
In the new substance class, these free spaces are arranged in such a way that the ions can move equally well in all directions. This is a result of the high degree of symmetry found in the crystals and is probably the cause of the "superionic lithium conductivity" which the TUM team has now been able to observe.
The synthesized powders are thus highly promising electrolyte candidates for future solid-state batteries. "Our basic research has the potential to accelerate the development of higher-performance batteries," Senyshyn concluded.
About the Author(s)
You May Also Like





