Medical Batteries Moving Forward
These highly specialized battery systems are benefitting from the development of lithium-ion batteries for laptops and EVs.
March 18, 2021
The medical battery business is a very conservative one. That’s a good thing, because you don’t want implanted pacemakers to fail catastrophically, or medical devices to perform unreliably. A whole gamut of battery chemistries is used for medical purposes, including nickel-cadmium (Ni-Cd), nickel-metal hydride (Nimh), and alkaline-manganese batteries. Lithium-ion batteries, the choice for EVs, personal electronics, and increasingly grid-scale electrical storage, are becoming popular.
Primary Batteries
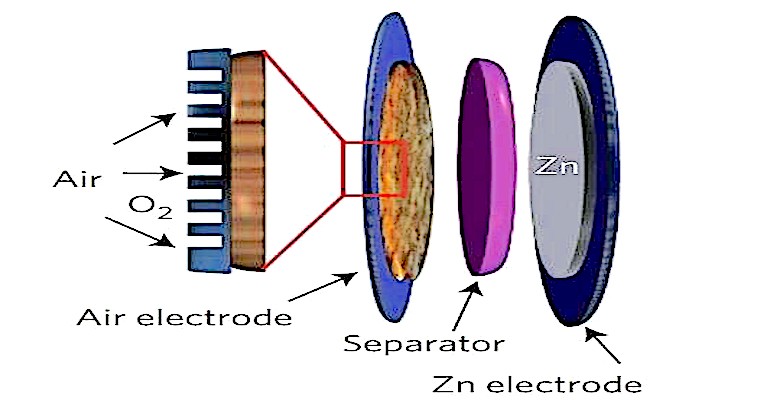
Non-rechargeable primary batteries are used in many medical devices. Zinc-air batteries, for example, are commonly found in hearing aids. These single-use batteries can provide high energy densities, are very light, and are relatively inexpensive to manufacture. The materials they contain are also relatively safe for disposal in the environment.
Here’s how they work. As a zinc-air battery discharges, zinc particles form a porous anode, which is saturated with an electrolyte. Oxygen from the air reacts at the cathode and forms hydroxyl ions which migrate into the zinc paste and form zincate (Zn(OH)4) while at the same time releasing electrons that travel to the cathode. The zincate decays into zinc oxide and water returns to the electrolyte. The water and hydroxyl from the anode are recycled at the cathode, so the water is not consumed. If the battery is kept sealed it can be stored for long periods. Removing the seal allows air to flow into the cell so that it can begin to produce electricity.
Primary batteries that are designed for implants are specifically designed to have an extremely long life and added stability, with wider margins for safety than typical commercial batteries. Battery maker EaglePicher supplies primary cells for implantable cardiac monitors and pacemakers and neurostimulators. It was also the first company with an FDA-approved implantable lithium-ion battery (2004).
Rechargeable Batteries
Medtronic is another company that has embraced lithium-ion batteries—since 2004, all of Medtronic’s rechargeable batteries are lithium-ion. The company uses inductive charging—the recharge system is sent home with the patient and is worn over the area where the device is implanted, recharging right through the skin.
In 2019, Dr. Marissa Caldwell, a battery research and technology scientist at Medtronic, told us, “For us, reliability is one of the first things that comes to mind. It means looking at reliability from all of the different facets, including what the requirements are from the device level and circuit level to how can we anticipate people using this device. What are the different recharge modalities? What are the possible failure modes that could happen? And all of this then gets wrapped up into the testing and modeling and designing. Most batteries that we design to be implantable are designed very conservatively. In order to build in this reliability, we do give up a lot of margins for, say, energy density. We will not be the most energy-dense cell on the market. But we will last longer and be more reliable.”

Rapidly Growing Market
The market for medical batteries is a rapidly growing one. First, medical technology is moving forward at a blinding pace, and most of the new devices that are under development require the mobility that a battery-powered system can provide. Secondly, there are a growing number of lifestyle-related health issues such as cardiac disease, diabetes, and obesity that require electronically powered monitors and other medical devices. Some of these devices regularly use hospital power from the wall socket but can switch to battery power when the patient needs to be moved from their room for testing or procedures.
North America dominates the market for medical batteries, primarily because of a broad application of medical technology in both the US and Canada. The large and increasing population in regions with growing economies, like Southeast Asia, China, and India, are expected to experience high growth in the medical battery market. As healthcare facilities in China, India, and the region improve, along with government funding for health care, the application of top-level medical technology will be a key driving force for medical batteries.
A Number of Players
Medical batteries, particularly the latest lithium-ion cells, come in a variety of shapes and sizes, powering everything from cardiac monitors and pacemakers, bioengineering devices, infusion pumps to external and implantable cardioverter defibrillators. Thin film and printed batteries are finding applications for miniaturized wearable and healthcare devices. There are many key players in the global medical batteries market. They include:
Siemens Ag
GE Healthcare
Maxim Integrated
Panasonic Corp
Texas Instruments
Quallion LLC
Stmicroelectronics N.V
Ultralife Corp
Electrochem Solutions
EaglePicher Technologies
Medtronic
Boston Scientific
Shenzhen A&S Power Technology Co., Ltd.
Although the global recession caused by the COVID-19 pandemic has caused a decline in the medical battery market, this is expected to only be temporary as health care spending rapidly increases into 2021 and beyond, due to a need for electrically powered and portable diagnostic and therapeutic devices.
Helping Move Forward
For a long time, the specialized nature of medical batteries has made them expensive to develop and manufacture. With the big push presently taking place to develop ever-improving, more powerful, and reliable batteries, particularly lithium-ion batteries, for applications like personal electronics and electric vehicles, the medical battery market will benefit. Improvements and cost reductions in raw material extraction and processing will help medical battery production on the manufacturing side. A push to recycle the materials from lithium-ion batteries will eventually help further reduce costs and environmental impacts. Better energy densities and power outputs from more compact battery cells, needed for longer range, better performing EVs, can only help when the same technologies find their way into medical batteries.
There will always be a lag between the latest technology and what is acceptable for medical batteries. There are a host of different medical-specific requirements from different medical working groups and especially the Food and Drug Administration (FDA). It’s important to prove that the batteries for medical devices will perform in the manner claimed. “It’s really hard to make a battery that’s going to last a long time, and it’s even harder to prove that it is going to last a long time,” summarized Caldwell.
Kevin Clemens is an engineering consultant who has worked on automotive and environmental projects for more than 40 years.
About the Author(s)
You May Also Like





