Air-Based Battery Technologies Promise Unprecedented Storage & Performance
As the industry strives for better battery capabilities, iron-air & lithium-air technologies are breaking barriers.
March 1, 2023

The demand for energy storage is growing at an unprecedented rate, as the world continues to rely heavily on renewable energy sources. This has led researchers to explore new and improved battery technologies that can provide higher energy density and longer lifetimes.
One place that is seeing particular interest is the air-based battery field. Recently, two air-based battery breakthroughs have shown significant potential to revolutionize energy storage. In this article, we’ll look at these breakthroughs to get a better understanding of the technology and its potential applications.
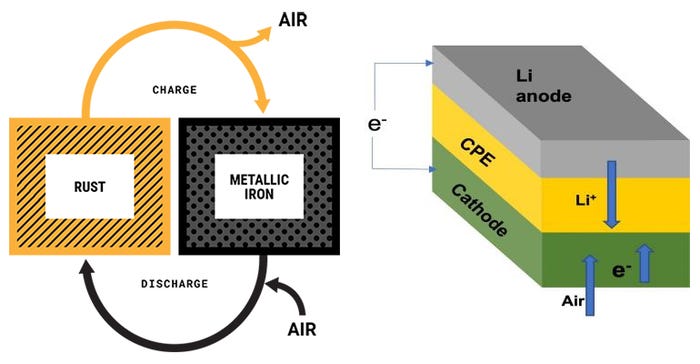
‘Rusty’ batteries
The first piece of battery technology news comes from Minnesota, where Great River Energy and Xcel Energy, two of Minnesota's largest utilities, are installing pilot iron-air battery storage projects to complement their renewable energy initiatives.
On a high level, the working principle behind iron-air batteries is that the battery takes in oxygen from the air and converts the iron to rust. Then, by applying an electrical current to the rust, the rust can be converted back into iron. The repeated process of rusting and unrusting the iron allows battery cells to charge and release electricity.
Unlike lithium batteries, iron-air batteries are capable of storing power for up to 100 hours and can be scaled to be more cost-effective. Although less efficient than lithium batteries, iron-air battery technology also has the benefits of being well-suited to extremely cold weather, recylcable, and sustainably sourced.
Great River Energy will install a 1.5 MW battery in Cambridge in 2022, while Xcel Energy will install a 10 MW battery in Becker. Minnesota hopes that the installation of iron-air battery technology can help create a more sustainable and clean future for the state. In pursuit of this, Great River Energy and Xcel Energy plan to use the batteries alongside renewable energy, to help manage how they put the energy those resources generate onto the grid.
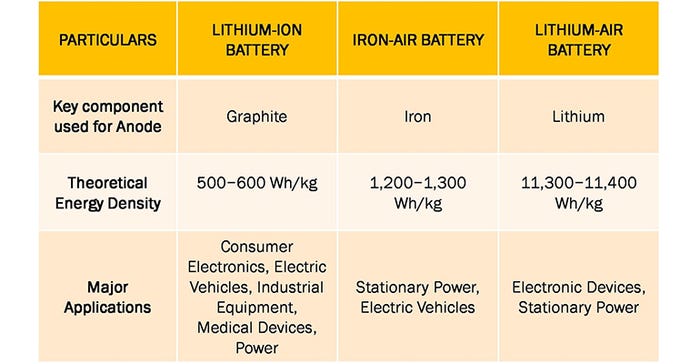
Lithium-air batteries
One emerging battery technology that is seeing heavy research is the lithium-air battery. Lithium-air batteries rely on a reaction between lithium and oxygen to generate electricity, which makes them highly efficient and potentially more energy-dense than traditional batteries. However, there are still several challenges that need to be addressed before the technology can be commercialized.
Now, a team of researchers at the Illinois Institute of Technology recently made headlines when they developed a new type of lithium-air battery that has the potential to provide significantly higher energy density than traditional lithium-ion batteries.
As described in the research paper, a novel aspect of this design was the use of a solid electrolyte made of a ceramic-polyethylene oxide composite, which is safer and more efficient than the liquid electrolytes used in traditional lithium-air batteries. The composite electrolyte was able to work at room temperature, which is also a significant improvement over previous designs that required high temperatures to function.
The resulting battery was able to sustain more than 1,000 recharge cycles with only a small drop in energy efficiency, and no impact on coulombic efficiency. According to the study, the solid-state electrolyte contributed around 75% of the total energy density, and the design has a lot of room for potential improvements surrounding minimizing the thickness to increase energy density.
While this is just a first step in the right direction, the researchers believe lithium-air batteries could eventually have significant impacts on transportation and heavy-duty vehicles such as airplanes, and trains.
About the Author(s)
You May Also Like





