EV Battery Electrolyte Formula Brightens Smiles at Argonne
New fluoride-containing electrolyte paves the way for better-performing, longer-lasting, and safer batteries for electric vehicles.
July 3, 2023

Argonne scientists have developed a new electrolyte for lithium metal batteries to power electric vehicles. It would greatly increase vehicle range, cost less than current designs, and enable an EV to run more safely, since the electrolyte is nonflammable. That would be enough to brighten the scientists’ smiles, but another aspect of the chemistry could do so literally: The electrolyte, like most toothpaste, contains fluoride.
An ingredient in many toothpastes is sodium fluoride, a compound of fluorine. It is added to protect teeth against decay. But compounds containing fluorine have other practical uses that might surprise you. Scientists at the US Department of Energy’s (DOE) Argonne National Laboratory have discovered a fluoride electrolyte that could protect a next-generation battery against performance decline.
“An exciting new generation of battery types for electric vehicles beyond lithium-ion is on the horizon,” stated Zhengcheng (John) Zhang, a group leader in Argonne’s Chemical Sciences and Engineering division.
The chemistries of non-lithium-ion batteries offer twice or more energy stored in a given volume or weight compared to lithium-ion. They could power cars for much longer distances and could even power long-haul trucks and aircraft one day. The expectation is that widespread use of such batteries will help address the problem of climate change. The main problem is that their high energy density declines rapidly with repeated charge and discharge.
One of the main contenders has an anode (negative electrode) made of lithium metal in place of the graphite normally used in lithium-ion batteries. It is thus called a “lithium metal” battery. The cathode (positive electrode) is a metal oxide that contains nickel, manganese, and cobalt (NMC). While it can deliver more than double the energy density possible with a lithium-ion battery, that outstanding performance rapidly vanishes within less than a hundred charge-discharge cycles.
The team’s solution involved changing the electrolyte, a liquid through which lithium ions move between cathode and anode to implement charge and discharge. In lithium metal batteries, the electrolyte is a liquid consisting of a lithium-containing salt dissolved in a solvent. The source of the short cycle-life problem is that the electrolyte does not form an adequate protective layer on the anode surface during the first few cycles. This layer, also called solid-electrolyte-interphase (SEI), acts like a guardian, allowing lithium ions to freely pass in and out of the anode to charge and discharge the battery, respectively.
The team discovered a new fluoride solvent that maintains a robust protective layer for hundreds of cycles. It couples a fluorinated component that is positively charged (cation) with a different fluorinated component that is negatively charged (anion). This combination is what scientists call an ionic liquid — a liquid consisting of positive and negative ions.
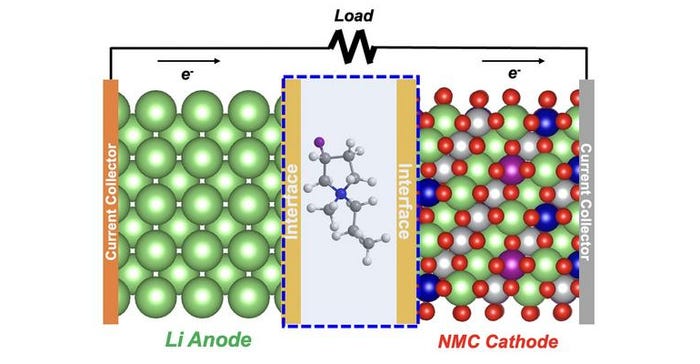
“The key difference in our new electrolyte is the substitution of fluorine for hydrogen atoms in the ring-like structure of the cation part of the ionic liquid,” Zhang stated. “This made all the difference in maintaining high performance for hundreds of cycles in a test lithium metal cell.”
To better understand the mechanism behind this difference at the atomic scale, the team drew upon the high-performance computing resources of the Argonne Leadership Computing Facility (ALCF), a DOE Office of Science user facility.
As Zhang explained, simulations on the ALCF’s Theta supercomputer revealed that the fluorine cations stick to and accumulate on the anode and cathode surfaces before any charge-discharge cycling. Then, during the early stages of cycling, a resilient SEI layer forms that is superior to what is possible with previous electrolytes.
High-resolution electron microscopy at Argonne and Pacific Northwest National Laboratory revealed that the highly protective SEI layer on the anode and cathode led to stable cycling.
The team was able to tune the proportion of fluoride solvent to lithium salt to create a layer with optimal properties, including an SEI thickness that is not too thick or thin. Because of this layer, lithium ions could efficiently flow in and out of the electrodes during charge and discharge for hundreds of cycles.
The team’s new electrolyte offers many other advantages as well. It is low cost because it can be made with extremely high purity and yield in one simple step rather than multiple steps. It is environmentally friendly because it uses much less solvent, which is volatile and can release contaminants into the environment. And it is safer because it is not flammable.
“Lithium metal batteries with our fluorinated cation electrolyte could considerably boost the electric vehicle industry,” Zhang stated. “And the usefulness of this electrolyte undoubtedly extends to other types of advanced battery systems beyond lithium ion.”
A paper on this research appeared in Nature Communications. In addition to Zhang, Argonne authors include Qian Liu, Wei Jiang, Jiayi Xu, Zhenzhen Yang, Doo-Joo Yoo, Krzysztof Z. Pupek, and Cong Liu. Other contributors include Chongmin Wang and Yaobin Xu from Pacific Northwest National Laboratory and Kang Xu from the U.S. Army Research Laboratory.
This work was supported by the DOE Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office. Computing time on the ALCF was awarded through DOE’s ASCR Leadership Computing Challenge.
About the Author(s)
You May Also Like




.jpg?width=300&auto=webp&quality=80&disable=upscale)
