A New Way to Make Powerful Solar Cells Might Drive a Reduction in Greenhouse Gasses
Researchers from NYU have streamlined a critical step in creating perovskite-based devices that also can benefit the environment.
June 13, 2021
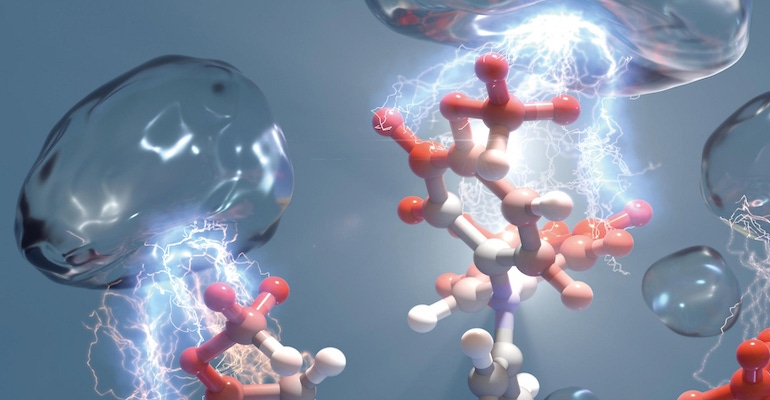
Solar energy may have environmental benefits because it creates a renewable energy source, but the process and materials used to make it are not necessarily ecologically friendly.
Now researchers have developed a way to make solar cells that not only creates more efficient devices but also can help capture greenhouse gases that cause environmental damage, they said.
The team from the NYU Tandon School of Engineering created the process for perovskite solar cells to alleviate a common bottleneck in a critical step of the process, which involves what’s called p-type doping of organic hole-transporting materials within the photovoltaic cells, researchers said.
Perovskite is an alternative to silicon being explored for future solar-energy-generating devices. However, they still are not on par with the performance and efficiency of more traditional solar cells—though scientists have made strides in recent years to achieve about 25.5 percent efficiency in the lab, from 3 percent in 2006, researchers said.
The novel process can help shorten device fabrication and processing time for perovskite solar cells as well as ultimately make them more stable, said Jaemin Kong, a post-doctoral associate who worked on the project, which was led by Associate Professor André Taylor. This can help them one day become a competitive commercial technology to silicon-based cells, Kong said.
Speeding Up the Process
Specifically, researchers improved the p-doping process involved in perovskite solar-cell production, which is achieved by the ingress and diffusion of oxygen into the hole transporting layer. Currently, this process is time-intensive, taking several hours to a day, limiting commercial production of perovskite cells.
The research team has a background in working with lithium-oxygen/air batteries and knew from experience that lithium-carbonate formation from exposure of oxygen electrodes to air is a challenge because it depletes the battery of lithium ions, thus destroying the battery capacity. However, in the case of the perovskite p-doping, researchers used carbon dioxide (CO2) instead of oxygen during the process to speed it up dramatically, they said.
In perovskite solar cells, doped organic semiconductors typically act as charge-extraction interlayers situated between the photoactive perovskite layer and the electrodes. How scientists typically dope these interlayers involves the addition of lithium bis(trifluoromethane)sulfonimide (LiTFSI), which is a lithium salt, to spiro-OMeTAD, a π-conjugated organic semiconductor widely used for a hole-transporting material in perovskite solar cells. The doping process is then initiated by exposing spiro-OMeTAD:LiTFSI blend films to air and light, researchers said.
Not only is this method extremely time-consuming, but it also is highly dependent on ambient conditions, researchers said. To speed it up, Taylor and his team created a fast and reproducible doping method that involves bubbling a spiro-OMeTAD:LiTFSI solution with CO2 under ultraviolet light, they said.
“In this Spiro doping reaction, we are actually exploiting lithium carbonate formation, which binds lithium and prevents it from becoming mobile ions detrimental to the long term stability of the perovskite solar cell,” Taylor explained in a press statement.
Benefits and Future Application
Researchers published a paper on their work in the journal Nature. They reported that the process rapidly enhanced electrical conductivity of the interlayer by 100 times compared to that of a pristine blend film, which is also about 10 times higher than that obtained from an oxygen bubbling process. The film treated with CO2 also resulted in stable, high-efficiency perovskite solar cells without any post-treatments, researchers said.
“Besides shortening the device fabrication and processing time, application of the pre-doped spiro-OMeTAD in perovskite solar cells makes the cells much more stable,” Kong explained in a press statement. “That’s partly because most of the detrimental lithium ions in the spiro-OMeTAD:LiTFSI solution were stabilized as lithium carbonates during the CO2 bubbling process.”
Moreover, the process has an environmentally friendly benefit—since it consumes quite a large amount of CO2 gas, it can be also considered for CO2 capture and sequestration studies in the future, Taylor said.
“At a time when governments and companies alike are now looking to reduce CO2 emissions if not de-carbonize, this research offers an avenue for reacting large amounts of CO2 in lithium carbonate to improve next-generation solar cells, while removing this greenhouse gas from the atmosphere,” he explained in a press statement.
The method also can be applied to other materials in p-type doping of other π-conjugated polymers, such as PTAA, MEH-PPV, P3HT, and PBDB-T, as researchers aim to push the boundary beyond typical organic semiconductors used for solar cells, Taylor said.
“We are hoping that this CO2 doping technique could be a stepping stone for overcoming existing challenges in organic electronics and beyond,” he said in a press statement.
Elizabeth Montalbano is a freelance writer who has written about technology and culture for more than 20 years. She has lived and worked as a professional journalist in Phoenix, San Francisco, and New York City. In her free time, she enjoys surfing, traveling, music, yoga, and cooking. She currently resides in a village on the southwest coast of Portugal.
About the Author(s)
You May Also Like