Is the Lithium-Ion Battery Dead?
Li-ion batteries will continue ruling the roost for now, but research points to sodium-ion batteries as the future.
January 4, 2024

The picture is emerging from the worlds of academia and industry research to consumer publications. Concluding a column titled “Sodium batteries offer an alternative to tricky lithium," The Economist in their Oct. 25,2023, issue concluded: “Perhaps the biggest disadvantage of sodium batteries is their late start. Lithium-ion batteries were first commercialized in the 1990s and have benefited from decades of investment. But the rest of the world is behind China on both fronts anyway. America and the European Union have announced enormous programs of green industrial subsidies. If they are determined to bankroll batteries, some of the pot should go to sodium.”
Lithium-ion batteries (LIB) are the dominant battery technology in the world and will continue thriving through the next decade, say many resources. They work well, are cost-effective in relation to their energy density and service life, and (spoiler alert) are much better for the climate than fossil fuels and fossil-based technologies, especially when it comes to transport.
Yet a looming lithium bottleneck remains. “The world cannot produce lithium-based batteries at the same rate as it wants to produce electric cars, and lithium deposits risk being depleted in the long term," says Rickard Arvidsson, professor of environmental systems analysis at Chalmers University of Technology in Gothenborg, Sweden. In addition, critical battery materials, such as lithium and cobalt, are largely mined in just a few places in the world, posing notable supply risks.
Arvidsson is lead author of “Prospective life-cycle assessment of sodium-ion batteries made from abundant elements,” published in November by the Journal of Industrial Ecology.

Rickard Arvidsson, professor of environmental systems analysis, Chalmers University of Technology, Gothenborg, Sweden. Credit: Chalmers University of Technology
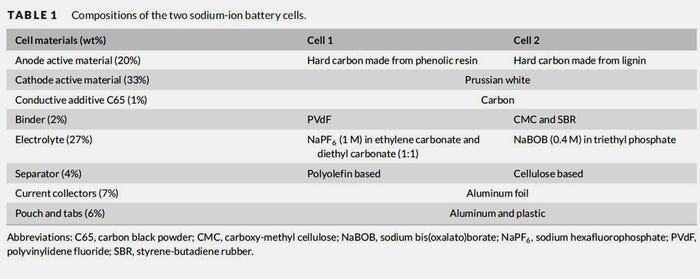
The university describes the study as a prospective life cycle assessment of two different sodium-ion battery cells where the environmental and resource impact is calculated from “cradle to gate,” meaning from raw material extraction to the manufacture of a battery cell. The functional unit of the study is 1 kWh theoretical electricity storage capacity at the cell level. Both types of SIB cells studied are mainly based on abundant raw materials. The anode is made up of hard carbon from either bio-based lignin or fossil raw materials, and the cathode is made up of so-called "Prussian white" (consisting of sodium, iron, carbon and nitrogen). The electrolyte contains a sodium salt. Since sodium-ion batteries are not currently produced at scale, production is modelled to correspond to a future, large-scale production. For example, the actual production of the battery cell is based on today's large-scale production of lithium-ion batteries in gigafactories.
Two different electricity mixes were tested, as well as two different types of so-called allocation methods – that is, allocation of resources and emissions. One where the climate and resource impact is distributed between coproducts based on mass, and one method where all impact is allocated to the main product (the sodium-ion battery and its components and materials).
Credit: CC BY-NC-ND DEED: Wickerts, S., Arvidsson, R., Nordelöf, A., Svanström, M., & Johansson, P. (2023). Prospective life cycle assessment of sodium-ion batteries made from abundant elements. Journal of Industrial Ecology, 1–14. https://doi.org/10.1111/jiec.13452
Earth-friendliest
The main conclusion from this study is that the SIB cells perform better than LIB cells regarding mineral scarcity for the cradle-to-gate system boundary. Another main conclusion is that SIB cells with Prussian white cathodes and LIB cells perform similarly for global warming and fossil resource scarcity given a cradle-to-gate system boundary. This is contrary to previous research that showed an SIB cell with Prussian white had almost twice the global warming compared to LIB cells: ca. 90 versus ca. 50 kg CO2 eq/kWh.
The two most important recommendations for improvements of the SIB cells and their production according to the study are that the gigafactory is powered by a fossil-free electricity supply, and that the hard carbon anode is produced from lignin instead of phenolic resin. Given a shift to renewable electricity for the gigafactory and a lignin-based anode, the electrolyte would be next up for having a large share of the impacts. Since none of the electrolytes considered in this study was clearly preferable over the other, further research into environmentally benign SIB electrolytes should be prioritized.
While sodium-ion batteries are not as yet produced at scale, although production is underway by a number of companies, an important step for improving the life-cycle model in this study would be to obtain large-scale SIB cell manufacturing data. In this study, large-scale manufacturing of LIB cells was adapted to SIB cells on a unit-process level, thus relying on a gigafactory originally designed for LIB cells. For example, SIB cells might require less dry-room facilities during production, which is known to be an energy-intensive part of LIB cell production.
For more information including a link to the entire study discussion, visit www.chalmers.se.
Note: Other than the happy coincidence of the exact same surname, there is no relationship between Ray Chalmers and Chalmers University of Technology. Ray does intend to order a hoodie.
About the Author(s)
You May Also Like





